Biotech Leaders

WCG Clinical | Insights
Virtual Clinical Trials: Best Practices in Moving Toward a Patient-Centric Research Model
Whitepapers
WCG Clinical | Insights
How to resolve irregularities in the documentation of informed consent?
Blog Posts
WCG Clinical | Insights
Can the IRB approve reimbursement of copays for routine costs in a clinical trial?
Blog Posts
WCG Clinical | Insights
Is IRB review required for survey results that may be published?
Blog Posts
WCG Clinical | Insights
Podcast: The Role of Expert Committees in Clinical Research with guest Jonathan Seltzer, MD, MBA, MA, FACC
Podcasts
WCG Clinical | Insights
The Role of Expert Committees: An Interview with Dr. Seltzer
Videos
WCG Clinical | Insights
Does the IRB need to review news stories?
Blog Posts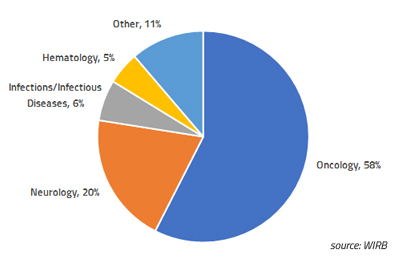
WCG Clinical | Insights
Single-Patient Expanded Access: WIRB experience in 2018
Blog Posts
WCG Clinical | Insights
Limited IRB Review: Are You Prepared for January 21st?
Blog Posts
WCG Clinical | Insights