Study Enablement
Overcome Barriers and Reach Study Goals Faster
Tackle study activation delays, training and safety challenges, and recruitment and retention complexities so you can run your studies more effectively and achieve your goals faster.

Run your studies more efficiently.
A successful clinical trial goes beyond a well-designed protocol – sponsors and CROs need to activate sites quickly, ensure site staff are well-trained and have the support they need, and recruit and retain the right participants to meet their study goals.
WCG’s Study Enablement offers tailored solutions to support sponsors and CROs in achieving optimal trial performance. Our comprehensive approach ensures that your trials are conducted efficiently and effectively, leading to better outcomes and more meaningful results. Partnering with WCG unlocks the full potential of your clinical trials, providing your study sites with the support they need for successful execution to drive groundbreaking medical advancements forward.
Our Study Enablement solutions include:
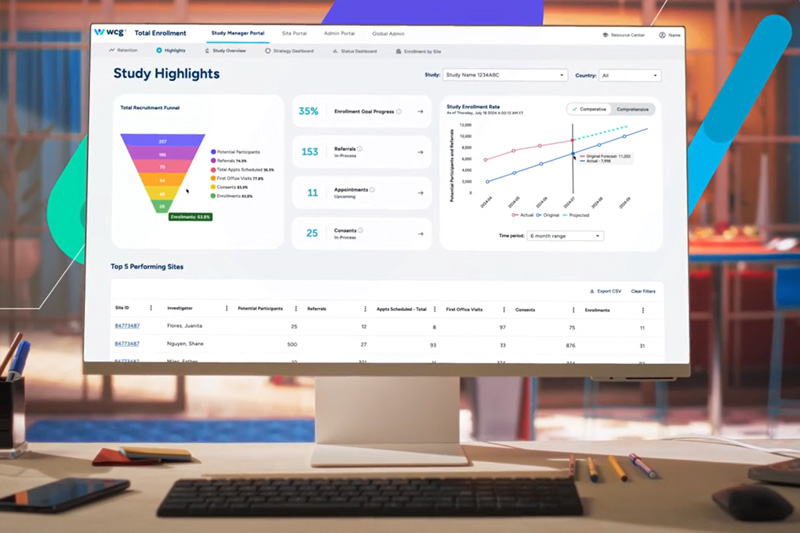
Recruitment & Retention
Secure and engage the right trial participants who align with your study’s criteria and will take your study over the finish line. WCG centralizes and simplifies participant identification, enrollment, retention, and documentation support, empowering you to devote your attention to what’s most important: the science, your sites, and your participants.
Clinical Trial Training
Streamline the training experience for study teams and sites with live, virtual, on-demand, and site initiation visit (SIV) meeting delivery with WCG InvestigatorSpace®, a technology that connects clinical trial training, safety reporting, rater qualification and regulatory document exchange with a single login.
Safety Letter Distribution
Manage critical and regulatory-required patient safety notifications to Principal Investigators through WCG InvestigatorSpace.
Site Network for Sponsors/CROs
Partner with a network of more than 470 clinical research site locations and 1,000+ PIs ready to conduct and accelerate your trials in nearly every therapeutic area.
Technologies designed to help enable more efficient and successful trials:
Why sponsors and CROs partner with WCG to optimize their studies:
Activate Sites Faster & Reduce Start-up Delays
Streamline site selection, budgets, contracting, and training so you can accelerate your study start-up timelines and start your trials sooner.
Improve Clinical Trial Training & Safety Reporting
Enhance training quality and effectiveness, streamline safety letter management, and simplify regulatory document exchange coordination all within a single login.
Simplify Participant Recruitment & Retention
Implement centralized recruitment and retention processes that equip your study sites with the necessary support, technology, and resources to conduct more successful trials.
Real Results
faster study start-up timelines when working with the WCG Site Network.
faster participant identification and enrollment.
month reduction in enrollment timelines.
reduction in the burden of site training.
Customer Stories
WCG has exceeded our expectations and have delivered remarkably during these past six months. When WCG got involved we were struggling more than I’ve ever experienced in any trial, and the manner in which WCG took ownership of this challenge was impressive.”
Top 5 Pharma Company
Working with the WCG Site Network helped us activate sites faster and streamline communication with our study sites so we could meet enrollment targets ahead of schedule!”
Mid-size Pharma Company
WCG has been extremely collaborative in providing recommendations for the development and implementation of our clinical trial training, which will ultimately lead to preserving data integrity in our studies.”
Top 10 Pharma Company
Enable Greater Study Success
Connect with WCG’s clinical trial experts to discover how we can help run your trials more efficiently and successfully.
Relevant Insights

WCG Completes Early Enrollment for Psoriasis & Atopic Dermatitis Study
Case Studies
Oversee investigator training and safety reporting for every site and study – with a single login
Blog Posts